Cardiovascular Pathology
14
Products
Abdominal Aortic Aneurysm
Clinical History This 70-year-old man with a history of mild gastro-oesophageal reflux suddenly developed severe upper abdominal pain radiating to the left shoulder tip. On examination, he was distressed and hyperventilating, with a pulse rate of 87/min and blood pressure 140/90 mmHg. Abdominal exam showed board-like rigidity and reduced bowel sounds. Emergency laparotomy revealed no ruptured viscus; the pancreas appeared normal, but an unruptured abdominal aortic aneurysm was noted. Endoscopy the next day identified a ruptured oesophageal ulcer, and a Celestin tube was placed. Despite treatment, he developed localized infection, pulmonary oedema, and congestion, dying 19 days after admission.Pathology The specimen shows the lower abdominal aorta, common iliac vessels, and proximal iliac arteries. A large 10 x 7 cm aneurysm lies below the renal arteries, extending to the aortic bifurcation. The aneurysm wall is severely thinned and partly lined by laminated thrombus, indicating chronicity. Recent thrombus formation is also evident. There is aneurysmal dilatation of the common iliac and left external iliac arteries. Multiple focally ulcerated atheromatous plaques are present at the upper aorta. No rupture is seen.Further Information An abdominal aortic aneurysm (AAA) is a localized enlargement of the abdominal aorta (diameter >3 cm or >50% larger than normal). Most AAAs are asymptomatic unless ruptured. Large aneurysms may be palpable, and sometimes abdominal, back, or leg pain occurs. Rupture causes sudden abdominal/back pain, low blood pressure, unconsciousness, and often death. AAAs mainly affect men over 50, especially smokers or those with high blood pressure or family history. Genetic conditions like Marfan’s and Ehlers-Danlos syndromes also increase risk. About 85% of AAAs occur below the kidneys, making them the most common aortic aneurysm.
Tetralogy of Fallot
Clinical HistoryA 21-month-old boy presented with a 2–3 month history of exhaustion and exertional dyspnoea. He had experienced several short episodes of acute breathing difficulty. On admission, he showed central cyanosis, mild finger clubbing and a harsh systolic murmur at the left sternal edge. Cardiac catheterisation confirmed Tetralogy of Fallot and severe pulmonary oedema. A Willis-Potts surgical anastomosis was performed. Unfortunately, the child died 12 hours post-operatively following acute dyspnoea and left lung consolidation.PathologyThe heart, viewed anteriorly, shows right ventricular hypertrophy and pulmonary outflow tract narrowing. The pulmonary valve is bicuspid and stenosed with visible endocardial fibrosis below. The aorta overrides a large ventricular septal defect (VSD) and communicates with the hypertrophied right ventricle. A probe inserted through the narrowed pulmonary trunk reaches the dilated left pulmonary artery and continues through the surgical anastomosis into the descending aorta. Posteriorly, the opened heart reveals a large atrial septal defect (ASD) and a second smaller ASD. Notably, the left ventricular wall is thinner than the right. Further InformationTetralogy of Fallot includes four key defects: 1. Ventricular septal defect, 2. Overriding aorta communicating with both ventricles, 3. Pulmonary stenosis, and 4. Right ventricular hypertrophy. Cyanosis typically appears early and its severity depends on the degree of right ventricular outflow obstruction. Some patients may survive untreated into adulthood, though surgical correction is now standard. Additional defects such as ASD may coexist. While the exact cause is unknown in many cases, genetic links include Down syndrome and DiGeorge syndrome.
Atrial septal defect
Clinical History A 10-year-old girl with a known congenital heart defect was admitted for surgical repair due to recent onset of cyanosis and cardiac failure. She was breathless, with blood pressure 105/60 mmHg and pulse 140/min. Examination revealed a loud heart murmur at the fourth left intercostal space near the sternum, elevated jugular venous pressure, and bilateral basal lung crepitations but no peripheral oedema. The defect was surgically repaired; however, the patient died following a sudden unexplained post-operative deterioration.Pathology The heart, viewed from the left side, showed a large 3.5 cm ovoid defect in the inter-atrial septum with only a small crescentic rim remaining. The left ventricle was small, while the right ventricle was hypertrophied, demonstrated by a thickened postero-lateral wall. The pulmonary artery was markedly enlarged, with the aortic arch above it and the left auricular appendage nearby.Further Information Atrial septal defect (ASD) is often asymptomatic early in life, even when large. Symptoms arise later due to reversal of the initial left-to-right shunt caused by increasing right ventricular hypertrophy and pulmonary hypertension. This leads to a right-to-left shunt, causing cyanosis, breathlessness, and ultimately congestive heart failure. Types of ASD include: - Secundum: Most common, located in the middle of the atrial septum. - Primum: Lower atrial septum, often with other congenital defects. - Sinus venosus: Rare, upper septum, usually with other heart anomalies. - Coronary sinus: Rare, wall between coronary sinus and left atrium is missing.The exact cause of ASDs is unclear. Some congenital heart defects are familial or linked to genetic conditions like Trisomy 21 (Down’s syndrome). Risk factors during pregnancy include infections (e.g., rubella), substance use (drugs, tobacco, alcohol), exposure to toxins, and maternal conditions such as diabetes or lupus.
Bicuspid Aortic Valve
Clinical History A 64-year-old woman presented with chest pain lasting 5 months, accompanied by breathlessness and wheezing for 4 months. On examination, she was dyspnoeic with an expiratory wheeze, left-sided crepitations, and signs of a right pleural effusion. Pulse and blood pressure were normal. A precordial systolic murmur and a heaving apex beat were noted in the 5th left intercostal space. There was no peripheral oedema. The patient died 4 days after admission.Pathology The heart was opened to reveal the left ventricle and valves. The aortic valve had only two cusps instead of three, with slight patchy thickening but otherwise normal valves. The coronary arteries, including the left circumflex artery, were widely patent. Dense pericardial fibrosis and adhesions on the posterior heart surface indicated constrictive pericarditis. Autopsy also showed ascites, a small cirrhotic liver, bilateral pleural effusions, and right lung collapse. Death was due to liver cirrhosis and failure, possibly linked to the pericarditis. The bicuspid aortic valve was an incidental finding.Further Information Bicuspid aortic valve is a common congenital anomaly, often asymptomatic until later in life. It predisposes to calcific aortic stenosis, usually in the 5th to 7th decades. The valve may be isolated or part of syndromes such as Tetralogy of Fallot. Fusion of two cusps results in unequal leaflet sizes, causing abnormal valve motion and turbulence, which increase the risk of aortic dilation, dissection, and calcification. Over time, this can lead to aortic stenosis or regurgitation, with symptoms like dyspnoea and reduced exercise tolerance. Diagnosis is confirmed by transthoracic echocardiography.
Acute Bacterial Endocarditis
Clinical History A 15-year-old boy presented with cough and sputum, then developed a hectic fever and chest pain days before admission in a comatose state. Examination revealed an early diastolic murmur at the aortic area radiating down the left sternal edge. Despite antibiotic treatment, he deteriorated rapidly and died. Blood cultures grew Staphylococcus aureus.Pathology The small heart specimen shows the left ventricle and valves. The non-coronary cusp of the aortic valve is ulcerated, perforated, and covered with friable vegetations. A perforation extends below this cusp into the right atrium above the tricuspid valve. Another aortic cusp is thickened. This represents acute bacterial endocarditis with aortic cusp and atrioventricular perforations.Further Information Acute bacterial endocarditis is a severe infection of the heart valves or endocardium, usually requiring prior damage to the endothelial lining for bacteria or fungi to adhere. Staphylococcus aureus is highly virulent and can infect even normal valves. The infection leads to platelet-fibrin aggregation forming vegetations, which grow due to coagulation activation and inflammation. These vegetations can embolize, spreading infection to distant organs. Risk factors include valvular disease (e.g., rheumatic or congenital heart disease), prosthetic valves, or previous cardiac procedures. Diagnosis involves clinical exam, blood cultures, and echocardiography (transthoracic and transoesophageal). Treatment consists of antibiotics, anticoagulants, and sometimes surgery.
Syphilitic Aneurysm
Clinical History A 61-year-old man presented with exertional anginal chest pain and dyspnoea, worsening over 6 years. On examination, he was cyanotic and tachycardic with a collapsing pulse. A swelling and thrill were noted on the right side of his neck. The apex beat was displaced inferolaterally, and a loud systolic and diastolic murmur was heard over the aortic area. Chest X-rays revealed cardiomegaly and a large rounded lesion in the right upper mediastinum continuous with the heart shadow, along with signs of cardiac failure. Blood tests were positive for anti-treponemal antibodies. Despite treatment, his condition deteriorated and he died from cardiac failure.Pathology The enlarged heart specimen includes the aortic arch and descending aorta. The ascending aorta was dilated up to 7 cm, with a large aneurysmal bulge measuring 11 x 13 cm. The aneurysm was opened, showing a wrinkled, scarred intimal surface with marked atheroma. The innominate, left common carotid, and subclavian arteries were displaced by the aneurysm. A 5 mm ridge-like thickening on the internal aneurysm surface marks the pericardial sac attachment. Adventitial vessels showed marked congestion. This is a syphilitic aneurysm of the aortic arch.Further Information Syphilis is a chronic infection caused by the spirochete Treponema pallidum, mainly sexually transmitted but sometimes congenital. Risk groups include sexually active individuals, intravenous drug users, HIV patients, and men who have sex with men. Penicillin remains the main treatment. Syphilis progresses in three stages: - Primary syphilis appears about 3 weeks after infection, with a painless chancre that heals spontaneously. - Secondary syphilis follows in untreated patients with systemic symptoms and characteristic rashes. - Tertiary syphilis develops years later and can cause cardiovascular syphilis, neurosyphilis, and gummatous lesions.Cardiovascular syphilis involves aortitis of the ascending aorta, leading to dilation, aortic valve insufficiency, and aneurysm formation from vasa vasorum endarteritis. Symptoms typically appear 15-30 years after infection. Neurosyphilis can cause headaches, vision loss, strokes, and cognitive decline. Gummatous syphilis causes nodular lesions in skin, bone, and mucosa, especially in HIV patients.
Right Ventricular Hypertrophy
Clinical History A 56-year-old woman with emphysema had a two-year history of worsening shortness of breath on exertion and recurrent bronchitis attacks. On examination, her blood pressure was 160/90 mmHg, pulse 96/min, and jugular venous pressure was elevated by 6 cm. The apex beat was impalpable, bilateral lung crepitations were present, and peripheral pitting oedema was noted. ECG showed a right heart strain pattern, and arterial blood gases revealed respiratory acidosis. Despite treatment, her condition steadily deteriorated and resulted in death.Pathology The heart, viewed anteriorly, showed a greatly enlarged and hypertrophied right ventricle. Otherwise, the heart appeared normal. This represents right ventricular hypertrophy (RVH) in a patient with emphysema.Further Information RVH commonly results from chronic lung diseases or structural heart defects. One main cause is pulmonary hypertension (PH), which raises pulmonary artery pressure and leads the right ventricle to compensate by hypertrophy. PH affects about 4 per million people globally, with RVH in approximately 30% of cases. Common causes of PH include chronic obstructive pulmonary disease (COPD), pulmonary embolism, and other restrictive lung diseases. RVH can also be caused by heart defects like tricuspid insufficiency, tetralogy of Fallot, ventricular septal defects, pulmonary valve stenosis, and atrial septal defects. Additionally, RVH is associated with abdominal obesity and elevated systolic blood pressure.
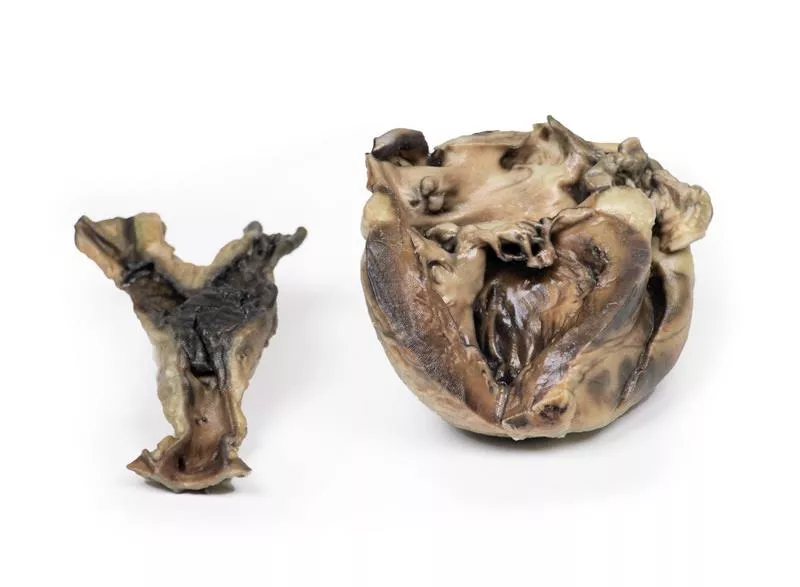
Congenital Pulmonary Stenosis
Clinical History This male child had a cardiac murmur detected at birth but remained well until 10 months of age. In the two weeks before hospital admission, he became lethargic, developed a mild cough, and experienced rapid weight gain. Swelling of the face, hands, and feet developed 10 days before admission. On examination, he appeared chubby, with a blood pressure of 90/59 mmHg. A fine thrill was present over the entire precordium, and a harsh systolic murmur was loudest in the pulmonary area. Peripheral oedema was noted. Despite treatment, his condition worsened, and death occurred due to congestive cardiac failure.Pathology The specimen is the child’s heart, viewed from the left. The pulmonary artery has been opened to show the upper surface of the pulmonary valve, which is malformed into a thickened conical diaphragm with a tiny 2 mm opening at the apex. A large post-stenotic dilatation of the pulmonary artery is visible. There is marked right-sided cardiac enlargement, with dilatation of the right atrium and auricle, as well as right ventricular hypertrophy. This case represents an example of pure pulmonary valve stenosis.Further Information Pulmonary stenosis accounts for approximately 7% of congenital heart defects. It can be isolated or part of more complex conditions like Tetralogy of Fallot. The severity of pulmonary stenosis ranges from mild to severe. While mild forms may remain undiagnosed for years, moderate stenosis may progress and severe cases cause substantial strain on the heart, leading to right ventricular hypertrophy and cardiac failure. Complications may include infective endocarditis and arrhythmias due to structural heart changes. Diagnosis is commonly made via echocardiography, though MRI and CT are also used. Surgical correction is now routinely possible.
Hydatid Disease Affecting the Heart and Aorta
Clinical HistoryAn 11-year-old girl with an 18-month history of hydatid disease underwent three craniotomies to remove 17 brain cysts. Additional cysts were later found in the kidneys, mesentery, and abdominal aorta at its bifurcation. Heart X-ray revealed a calcified cyst, and during its surgical removal, the patient deteriorated and died. A dead hydatid cyst was found in the left ventricle.PathologyThe heart, opened to expose the left ventricle, shows significant left ventricular hypertrophy and an abnormal channel through the posterior mitral valve cusp into the left atrium, surrounded by fibrous tissue. Surgical sutures are visible. The abdominal aorta at the iliac bifurcation is filled with a large antemortem clot. Hydatid cysts are found in both the aortic wall and the ventricular-atrial channel, displaying characteristic layered structures with daughter cysts and surrounding granulomatous reactions.Further InformationHydatid disease is caused by the tapeworm Echinococcus granulosus, transmitted via dog feces. Humans may ingest the eggs, leading to cyst development, most commonly in the liver and lungs. Cardiac involvement is rare (0.5–2% of cases) but can be fatal. Cysts typically form subepicardially and may rarely rupture into the pericardium, causing tamponade or other complications.
Hypertrophic Subaortic stenosis
Clinical HistoryA 42-year-old American tourist was found dead in his hotel room. A coroner’s autopsy was conducted to investigate the cause of death.PathologyThe heart specimen shows a longitudinal section through both ventricles and the interventricular septum. The most striking feature is the marked thickening of the septum and left ventricular hypertrophy. The aortic and mitral valves appear normal. The enlarged septum narrows the left ventricular lumen. DiagnosisIdiopathic hypertrophic subaortic stenosis, also known as hypertrophic cardiomyopathy. Further InformationSubaortic stenosis is typically acquired, arising from an abnormality in the left ventricular outflow tract (LVOT). Turbulent blood flow in this area may lead to progressive thickening and fibrosis, causing increased resistance and eventual hypertrophy of the left ventricle. While mild cases are often asymptomatic, more advanced disease can present with exertional dyspnoea or syncope. A systolic murmur may suggest the diagnosis, which is typically confirmed via echocardiography. In severe cases, surgical correction of the obstruction is the treatment of choice.
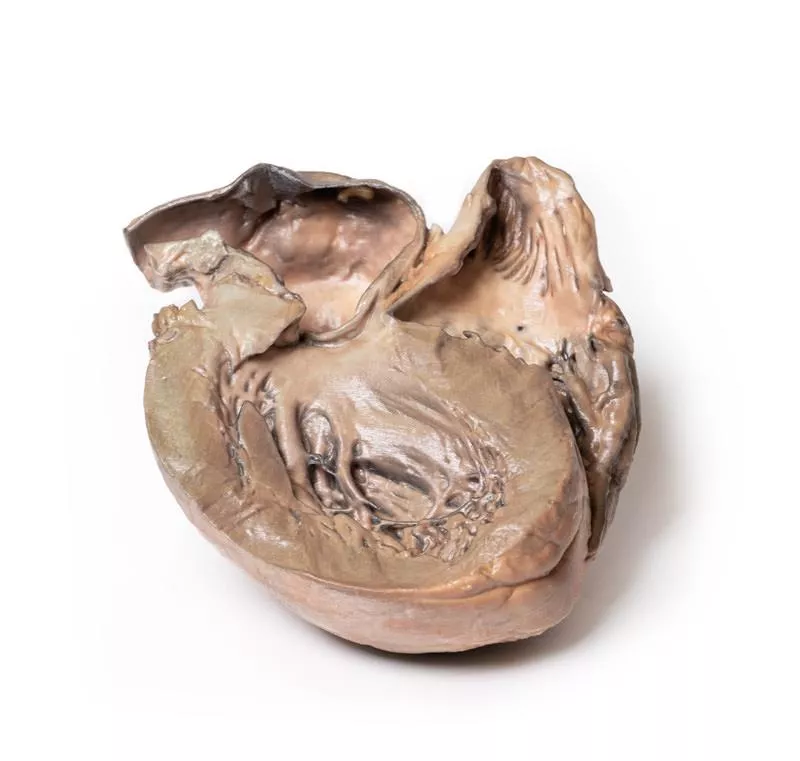
Rheumatic endocarditis
Clinical HistoryA 52-year-old woman presented with increasing dyspnoea. Her history included childhood fever with migratory joint pains following a sore throat. On examination, she was cyanotic, in atrial fibrillation, had elevated jugular venous pressure, a pan-systolic murmur at the apex, hepatomegaly, and dependent oedema. She received digoxin, furosemide, and penicillin but died after cardiac arrest.Pathology The opened heart shows the left atrium and ventricle. The mitral valve is thickened. The left atrial wall is lined with blood and fibrin, and the appendage is filled with clot due to atrial fibrillation. A mural thrombus and irregular endocardial thickenings—known as MacCallum’s plaques—are evident at their characteristic location. Further InformationThis presentation strongly suggests rheumatic fever, an autoimmune reaction following untreated streptococcal throat infection. It often involves the heart, joints, skin, and brain. Repeated cardiac involvement can lead to chronic rheumatic heart disease, most commonly damaging the mitral valve. Fibrosis causes stenosis, often with characteristic Aschoff nodules and, over time, dilatation of the left atrium. This can trigger atrial fibrillation and mural thrombus formation, ultimately resulting in severe heart failure.
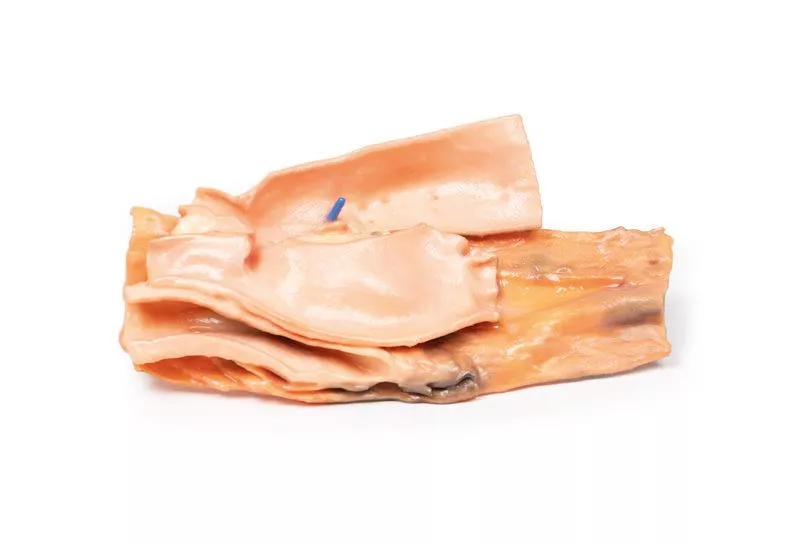
Traumatic Oesophageal-aortic fistula
Clinical History A woman swallowed a chop bone during lunch and later collapsed, suffering a massive haematemesis. At laparotomy, the stomach was filled with fresh blood, but the cause was not identified. She died the next day. Necropsy revealed a communication between the aorta and the oesophagus.Pathology The specimen includes the distal trachea, aortic arch (opened coronally and viewed anteriorly), and oesophagus (opened longitudinally). The oesophageal mucosa is ulcerated and haemorrhagic. A small probe shows a fistula connecting the oesophagus with the posterior wall of the thoracic descending aorta.Further Information Though this case was caused traumatically, aorto-oesophageal fistulas can also arise non-traumatically from aortic aneurysms compressing the oesophagus, gastrointestinal cancers, or erosion of aortic grafts into the digestive tract. Such fistulas can occur anywhere along the aorta. They are life-threatening and commonly present as gastrointestinal bleeding, ranging from minor bleeding to massive haemorrhage causing haemodynamic collapse. Symptoms include melaena, frank bleeding in stools, or haematemesis as in this case. Smaller fistulas may cause malaise or limb ischaemia due to reduced blood flow. Diagnosis can be challenging and depends on patient stability. Stable patients may be evaluated by endoscopy or CT angiography, while unstable patients often require urgent laparotomy and blood transfusions.
Ruptured Thoracic Aortic Aneurysm
Clinical History No clinical details are available for this specimen.Pathology The heart is shown from the posterior aspect, displaying both ventricles. A prominent saccular dilatation of the ascending thoracic aorta is present, featuring several atherosclerotic plaques. The aorta is ruptured posteriorly, indicated by dark staining. Both ventricles are hypertrophied, while the coronary arteries and aortic and tricuspid valves appear normal. This represents a ruptured aneurysm of the ascending aorta.Further Information Dilation of the ascending aorta is often an incidental finding on transthoracic echocardiography. The thoracic aorta consists of three parts: ascending, arch, and descending. The ascending aorta begins just beyond the aortic valve and ends before the brachiocephalic trunk, measuring about 5 cm in length. It includes the aortic root (with coronary sinuses and sinotubular junction) and the tubular ascending aorta. Over 50% of thoracic aortic aneurysms occur here, affecting either the root or tubular segment. An aneurysm is a localized dilation of the aorta exceeding 50% of the expected diameter (observed/expected ratio = 1.5) and differs from ectasia, which is a diffuse dilation less than 50%. The incidence of ascending thoracic aortic aneurysms is about 10 per 100,000 person-years. Reference: Saliba et al. (2015). Int J Cardiol Heart Vasc. 6: 91–100.
Calcified Aortic Valvular Stenosis Bicuspid Aortic Valve
Clinical HistoryThere is no available clinical history for this specimen.Pathology This specimen is a partial horizontal slice, approximately 1.5 cm thick, taken through the plane of the left atrium. The smooth internal surface of the atrium, the left auricular appendage, and a section of the left ventricle are visible on the inferior aspect. On the superior side, the pulmonary trunk and part of the pulmonary tricuspid valve are visible, along with the aorta and an abnormal bicuspid aortic valve. This valve shows calcified thickening at the opposing margins. Additionally, there is calcification on one of the pulmonary valve cusps. Further InformationA bicuspid aortic valve is the most common congenital heart defect, often undetected until adulthood. It can lead to aortic valve stenosis, which may range from mild to severe. Symptoms—when they occur—include heart murmur, angina, shortness of breath, palpitations, and signs of heart failure such as fatigue and swollen ankles. The narrowed valve forces the left ventricle to work harder, which can lead to ventricular thickening, enlargement, and eventual heart failure. Calcium buildup contributes to valve stiffening, especially in those with bicuspid valves, further narrowing the lumen and restricting blood flow.
Human body replicas to improve teaching!
Erler-Zimmer's groundbreaking Anatomy Series features a unique and unrivalled collection of colourised human body replicas specifically designed to enhance teaching and learning. This premium collection of highly accurate human anatomy has been created directly from radiological data or real specimens using the latest imaging techniques. The 3D Human Anatomy Series offers a cost-effective way to meet your specific teaching and demonstration needs across the curriculum in medicine, health sciences and biology. A detailed description of the anatomy represented in each 3D printed specimen is included. What are the advantages of the Monash 3D Anatomy Series compared to plastic models or real human plastinates?
Each body replica has been carefully developed from selected radiological patient data or dissected human bodies of the highest quality, chosen by a highly skilled team of anatomists at Monash University's Human Anatomy Teaching Centre, to represent clinically important areas of anatomy in a quality and detail not possible with conventional models - it is real anatomy, not stylised. Each body replica has been rigorously checked by the highly qualified team of anatomists at Monash University's Human Anatomy Teaching Centre to ensure the anatomical accuracy of the final product. The body replicas are not real human tissue and are therefore not subject to any restrictions on transport, import or use in educational institutions that do not have permission to use cadavers. The
exclusive 3D Anatomy Series avoids these and other ethical issues that arise when dealing with plastinated human remains.